|
Each of these supergiant, iron-containing stars only lives for a brief while before violently blasting as a supernova, scattering iron into space and onto rocky planets like Earth. It is produced abundantly in the core of massive stars by the fusion of chromium and helium at extremely high temperatures. Iron occurs naturally in the known universe. Commonly, iron uses two (oxidation state +2) or three (oxidation state +3) of its available electrons to form compounds, although iron oxidation states ranging from -2 to +7 are present in nature. As with other transition metals, a variable number of electrons from iron’s two outermost shells are available to combine with other elements. In February, we have selected iron, the most abundant element on Earth, with chemical symbol Fe (from the Latin word “ferrum”) and atomic number 26.Ī neutral iron atom contains 26 protons and 30 neutrons plus 26 electrons in four different shells around the nucleus. 1 and dissected hydrogen’s role in oxidation-reduction reactions and electrochemical gradients as driving energy force for cellular growth and activity. 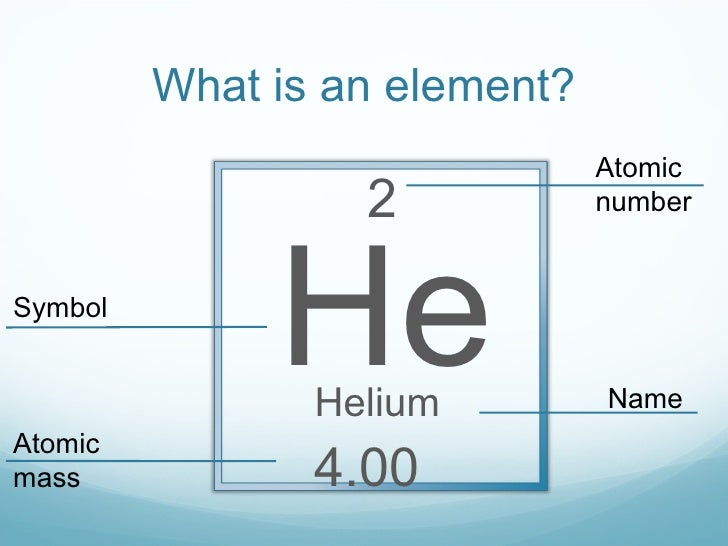
In iron-deficiency anemia, the heart works harder to pump more oxygen through the body, which often leads to heart failure or disease.We are celebrating the 150th anniversary of Mendeleev’s periodic table by highlighting one or more chemical elements with important biological functions each month in 2019. Each monomer contains a heme group in which an iron ion is bound to oxygen. Because its 2n shell is filled, it is energetically stable as a single atom and will rarely form chemical bonds with other atoms.Hemoglobin is a tetramer that consists of four polypeptide chains. For instance, lithium ( Li \text Ne start text, N, e, end text ), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1 s 1, s orbital and eight fill the second shell-two each in the 2 s 2s 2 s 2, s and three p p p p orbitals, 1 s 2 1s^ 2 1 s 2 1, s, squared 2 s 2 2s^ 2 2 s 2 2, s, squared 2 p 6 2p^6 2 p 6 2, p, start superscript, 6, end superscript. Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. 
This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. 
The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
